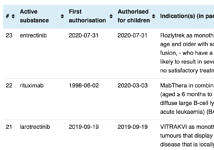
A continually updated list of centrally authorised products (updated 2026-01-14)
Ralf Herold's blog on progress of medical research and medicine development

A continually updated list of centrally authorised products (updated 2026-01-14)
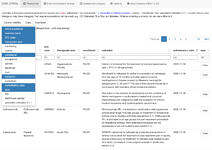
Interactive webapp about centrally authorised medicines and their assessments (EPARs), updated at least once a week
The post presents a readily-usable tool with a permissive licence for CTIS2, EUCTR, CTGOV2 and ISRCTN with examples addressing research questions (updated 2026-01-25)
A list of tools such as biomarkers, simulation approaches, new endpoints, novel methods, commented by regulatory agencies, updated daily

Videos, interviews, publications and presentations by Ralf Herold to promote progress for children and adults with cancer (updated 2024-11-30)
 Many are relevant to be studied in children with cancer (updated 2026-01-24)
Many are relevant to be studied in children with cancer (updated 2026-01-24)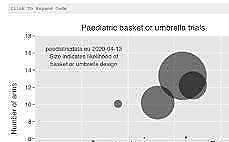 Clinical trials with children are evolving and improving (updated 2020-04-13)
Clinical trials with children are evolving and improving (updated 2020-04-13)Distributed and diverse public information is combined by this web service and shared in APIs for deepening understanding and improving of medicine development and availability (updated 2020-09-12)